
Nurturing life’s potential with bottled brilliance
Optimise embryo development with Vitrolife’s all-inclusive IVF media suite
We believe that medically assisted reproduction should be fortified by cutting edge science and an unparalleled arsenal of tools. Our portfolio of IVF media is a testament to this belief – and a reflection of our commitment to creating the most supportive environment for embryo development.
Since we started in 1994, every product in our IVF media portfolio has been crafted with utmost precision to form a toolset that embodies the highest standards of consistency and excellence. As new breakthroughs in medically assisted reproduction emerge, our portfolio evolves to ensure that you are always equipped with the most efficient tools available.
Performing IVF procedures is a significant responsibility that requires a solid foundation of reliability, consistency, and scientific precision. This is precisely what our IVF media portfolio delivers. Year to year. Batch to batch.
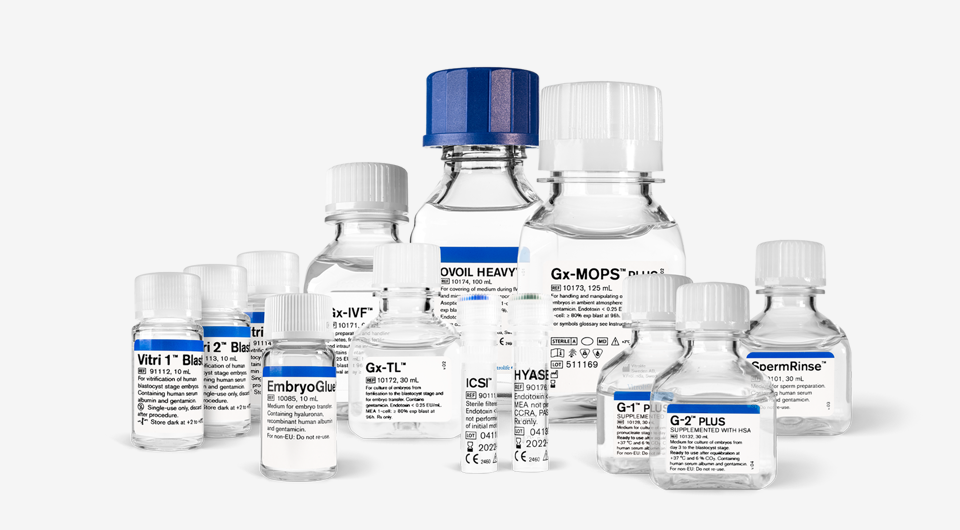
A pursuit of excellence in IVF media
In the complex world of IVF, we are constantly faced with challenges that demand precision, reliability, and an in-depth understanding of reproduction. Vitrolife’s IVF media has been meticulously designed to address these challenges.
Achieving high pregnancy rates pose significant challenges for medically assisted reproduction centers. Gametes and embryos are exceptionally vulnerable to oxidative stress. When cultured and handled in vitro, they lack the natural detoxification mechanisms of the human body.
By harnessing the power of scientific advancements and research, we have crafted a reliable media portfolio that strives to mimic nature’s intended environment for the embryo, and to shield against in vitro stresses.
Go to full assortmentFactors influencing pregnancy rates
Success in the laboratory hinges on meticulous attention to detail. Even minor disruptions in the environment can diminish an embryo’s ability to implant and subsequently reduce the likelihood of a live birth. Within the in vitro setting, gametes and embryos commonly face challenges.
To counter these, the chosen media should act as a buffer against external disturbances and optimise every embryo’s chance of developing into a baby. From the moment of sperm collection and oocyte retrieval to the point of transfer, gametes and embryos depend on media developed to nurture biological processes and streamline laboratory workflows.
Challenges in the in vitro setting
- Deviations in pH and temperature.
- Elevated osmolality.
- Exposure to reactive oxygen species
(ROS) stemming from suboptimal
plasticware, light and oxygen.


Vitrolife media in the IVF laboratory
Within the human body, embryos develop in an optimal microenviroment within the female reproductive tract, shielded from external stressors. The ideal media composition must be tailored to gametes and embryos, as well as adapted to the in vitro laboratory processes and equipment, which are changing over time. This necessitates precise control of pH, osmolality, and temperature, along with the avoidance of any form of toxicity.
“By working through each of the processes together in the laboratory we combined the Vitrolife labware and media into a, for us, optimised way of working. From this day on we have seen huge improvements, on all important quality KPIs. The best with the system is its consistency. There are marginal gains, and it is a really robust system, tailored for our needs.”
- Qamar Waylat, Lab Manager, Birmingham Women’s Fertility Centre
The research of Professor David Gardener
Distinguished Professor, School of BioSciences, University of Melbourne and Scientific Director, Melbourne IVF
The evolution of our products has been shaped by years of in-depth media research and invaluable interactions with customers worldwide. The research of Professor David Gardener has also led to pivotal discoveries that are now integral components of our products:

1998
Sequential media
The development of blastocyst culture and transfer, marked by the creation of sequential culture media, G1 and G2 1, was a significant milestone in human IVF. These advancements substantially boosted the implantation rate and paved the way for single embryo transfers worldwide 2, 3.
2003
The inclusion of hyaluronan in culture media and the birth of EmbryoGlue
Discovering that the female reproductive tract contains not just albumin but various macromolecules, we explored the role of hyaluronan in supporting embryo development and viability. This research laid the groundwork for EmbryoGlue 4, 5. Today, hyaluronan is a vital ingredient in Vitrolife’s culture, transfer, and cryo media.
2010
The development of the Rapid-i Vitrification System
Vitrolife’s vitrification media is designed to operate at physiological temperature to preserve spindle integrity. Working at 37°C shortens the vitrification time and limits exposure to cryoprotectants like propanediol and ethylene glycol. A significant achievement was the design of a closed vitrification system, avoiding direct contact with liquid nitrogen, supporting oocyte and embryo safety 6, 7, 8.
2015
G-TL for time-lapse
The rise of time-lapse technology and the growing demand for uninterrupted culture led to the creation of the medium G-TL. This medium was specifically designed to ensure that when amino acids undergo deamination, the resulting ammonium levels remain non-toxic to embryos 9.
2018
Pioneering an antioxidant system
Oxidative stress is an inevitable outcome of metabolism and in vitro culture. Our research resulted in an antioxidant system, Gx Media, capable of effectively mitigating oxidative stress in gametes and embryos, in both ambient and reduced oxygen 10, 11, 12.
Use the complete system for optimal results
While time-lapse technology, preimplantation genetic testing and vitrification offer new treatment possibilities, they also present new challenges to the way we work. Vitrolife media is designed as a serie, with each product offering support to the specific developmental stage and treatment it addresses. Always crafted with your practice in mind, our media prioritises three core values: safety, viability, and clinical outcome.


Safety
Consistent results are achieved by ensuring a stable and non-toxic environment. We are committed to providing you with thoroughly developed products and validated protocols. For instance, achieving success with vitrification and ensuring cell viability requires balancing cryoprotectant concentration, exposure time, volume, and temperature. Our protocols have been validated and approved by regulatory authorities, assuring confidence in our processes.
One of the most critical components in a culture media system is oil13. Vitrolife’s oils, OVOIL and OVOIL HEAVY, provide a protective environment for your culture systems.
Learn more about the safety of OVOIL
Viable embryos
Reactive oxygen species are ubiquitous throughout any given IVF cycle, posing a significant threat to your embryo culture system. To keep embryos out of harm’s way, we have developed Gx Media. The unique triple antioxidant combination in Gx Media, consisting of acetyl-L-carnetine, alpha-lipoate and acetyl- L-cysteine, form a protective barrier against oxidative stress and support embryo viability.
The triple antioxidant combination can be found in three familiar products, now becoming Gx Media, including Gx-MOPS PLUS, Gx-IVF and Gx-TL. These products support a wide range of laboratory procedures, including sperm preparation, oocyte pick-up, fertilisation by IVF and ICSI, as well as culture and transfer.
Learn more about Gx Media
Clinical outcome
When speaking about clinical outcomes, EmbryoGlue holds a special position. It was developed exclusively for transfer and is the only existing transfer medium with a proven implantation-enhancing effect 21.
It is suitable for transferring embryos at all developmental stages, from cleavage-stage embryos to blastocysts. Using EmbryoGlue for embryo transfers can notably increase both clinical pregnancy and live birth rates 21.
Learn more about EmbryoGlue
Superior quality and support
Quality yields results. Our commitment to quality control and clinical support ensures stability in your culture system.
The Mouse Embryo Assay (MEA) is our most important biological assay, the key to ensuring that we deliver safe and consistent products. Designed to detect toxicity and pinpoint suboptimal raw materials, contact materials, media, and instruments, our MEA is applied comprehensively. We rigorously screen the majority of materials used during manufacturing and test our finished products before they can be released.
Learn more about how we perform MEA

Advancing clinical excellence together
Products must be adapted to fit the unique needs and realities of your laboratory and clinic. While the Vitrolife product portfolio is designed for comprehensive utility, there may be times when customisation is beneficial. Peer discussions often offer valuable insights. This is where the Vitrolife Academy steps in - a global network of Vitrolife IVF specialists dedicated to providing clinical support to IVF clinics. The Academy emphasises the holistic product and procedural solutions that Vitrolife offers, with the goal of enhancing efficiency and improving clinical outcomes.
Learn more about Vitrolife AcademyA customer experience story about clinical customisation
Embark on a journey with Dr. Daniela Nogueira, a seasoned IVF expert with over 30 years of global experience. From Belgium to the UAE, her career has touched three continents, emphasising the importance of customisation in fertility treatments. Discover her insightful perspective on Vitrolife Group, a pioneer in IVF media, and how our scientifically sound approach transformed her clinic's results. With over two decades of trust, Dr. Nogueira's endorsement solidifies Vitrolife Group's position at the pinnacle of IVF product excellence.
Learn more
Training at your convenience – online and onsite
Vitrolife Academy focuses on providing workshops, webinars and educational material to our customers and we strive to help IVF laboratories to work in the most optimal way. Vitrolife Academy offers online training on demand, a complement to our other courses and workshops, to deepen your knowledge.
Request account
References
- Gardner, D. K., & Lane, M. (1997). Hum Reprod Update, 3(4), 367-382, Article G-1, G-2.
- Gardner et al. (1998). Hum Reprod., dec, 13(12), 3434-3440, Article G-1, G-2, SA-Solution, ICSI.
- Gardner et al. (2000). Fertil Steril, 73(6), 1155-1158.
- Gardner et al. (1999). Hum Reprod, 14(10), 2575-2580, Article.
- Schoolcraft et al (2002). Fertil Steril, 76(Suppl 3), S5, O-11, Article G-1, G-2, G Series (v3), EmbryoGlue.
- Gardner et al. (2007). Theriogenology, 67(1), 64-72, Article -. https://www.sciencedirect. com/science/article/abs/pii/S0093691X0600505X.
- Larman & Gardner. (2011 Fertil Steril., mar 15, 95(4), 1462-1466, Article Rapid-i, G-1, G-2, HSA-Solution, RapidVit Cleave, RapidWarm Cleave (animal data).
- Larman et al. (2006). Reprod Biomed Online, 12(1), 66-69, Article OVOIL, G-MOPS (animal data).
- Hardarson et. al. (2015). Fertil Steril, 104(6), 1452-1459 e1454, Article Primo Vision, Ovoil, G-TL, G-1 PLUS, G-2 PLUS.
- Gardner & Truong (2017). Human Reprod, P-248, Article.
- Truong et al. (2022). Reproductive BioMedicine Online, 44(3), 393-410.
- Truong et al. (2016). Hum Reprod, 31(7), 1445-1454, Article G-MOPS PLUS, Ovoil, EmbryoScope, G-1 (v5), G-2 (v5) (animal data).
- Cairo Consensus Group, ‘There is only one thing that is truly important in an IVF laboratory: everything’ Cairo Consensus Guidelines on IVF Culture Conditions. Reprod Biomed Online, 2020. 40(1): p. 33-60.
- Mouse Embryo Assay for Assisted Reproduction Technology Devices. Doc issue Jan 5 2021, https://www.fda. gov/media/127874/download
- Gurner K H et al. (2023). Comparison of OVOIL and OVOIL HEAVY in the clinical laboratory on fertilisation, blastocyt utilisation and transfer outcomes). Poster Aspire 2023.
- Gardner et al. (2020). Reprod Biomed Online, 40(5), 637-644, Article G-MOPS PLUS, G-IVF PLUS, G-1 PLUS, G-2 PLUS.
- Kelley et al. (2023). O-235. Hum Reprod, 38(Supplement_1). https://doi.org/10.1093/humrep/dead093.285.
- Mizumoto et al. (2021). Hum Reprod, 36(deab130.220), Article G-TL, Gx-TL.
- Hardarson et al. (2018). Fert Steril, 110(4), e51, O-115, Article G-TL, G-TLX.
- Ueno et al. (2021). Reprod Sci, Article Gx-IVF, Gx-MOPS, SpermGrad, EmbryoScope PLUS, EmbryoSlide, EmbryoViewer Software, Gx-TL, OVOIL.
- Heymann et al. (2020). Cochrane Database Syst Rev, 9, CD007421. 22. Urman et al. (2008) Fertil Steril (90):3.