G-1™ PLUS
G-1™ PLUS provides the carbohydrates, amino acids and hyaluronan needed to support the embryo during the first critical stages of development.
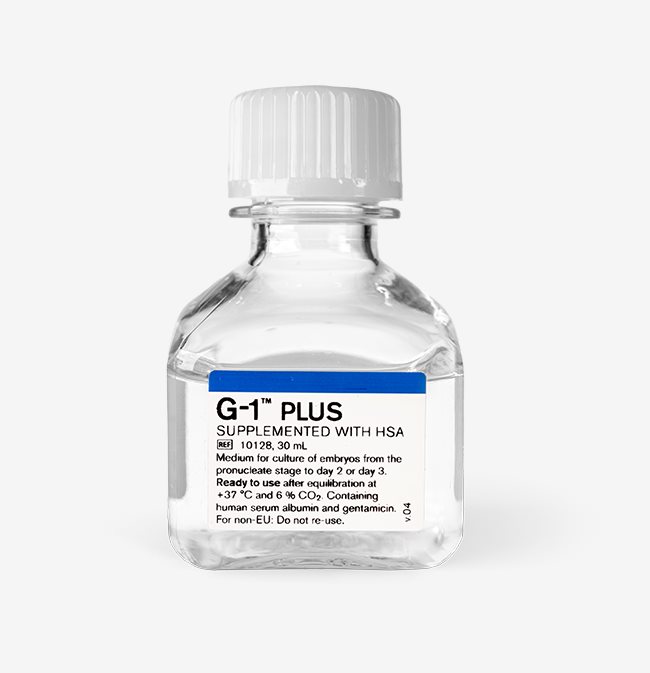
Description G-1™ PLUS is a bicarbonate buffered media containing hyaluronan. It also contains human serum albumin (5 mg/ml).
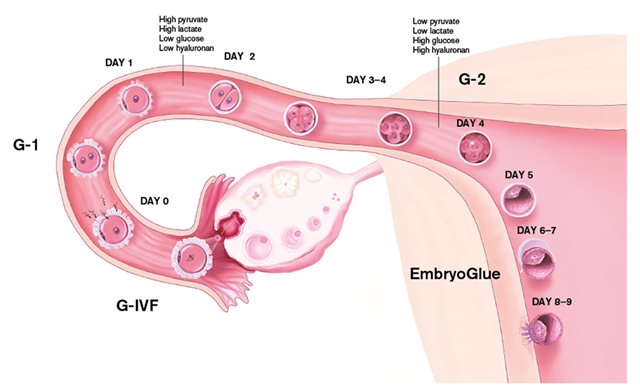
Purpose Medium for culture of embryos from the pronucleate stage to day 2 or day 3.
Application For use after equilibration at +37°C and 6 % CO2.
Storage Store dark at +2 to +8°C
Important nutrients supports the embryo
G-1 PLUS is a media for culture of human embryos from after fertilisation until the 8-cell stage. G-1 PLUS is supplemented with human serum albumin, HSA, and is ready to use. It contains amino acids to support embryo viability and hyaluronan to improve development and cryosurvivability and to facilitate implantation. It also contain lipoic acid as antioxidant to protect the embryo against reactive oxygen species during culture in vitro.
G-Series - confidence at each step
Each product in the G-Series™ is developed to resemble conditions in the female reproductive tract and fulfil embryo requirements.
Support Documents
Documents Language

Package inserts
English
Package insert G1 PLUS - Multi-language
Document and Ref: REF 26080, version 10

Safety data sheets (SDS)
English
SDS G-1 PLUS
Document and Ref: Document and Ref: Date Revised : 04/16/2025.Revision No : 6. Safety data sheet for G-1 PLUS

Scientific summaries
English
Optimised clinical outcomes with blastocyst culture and cryopreservation using Vitrolife G-Series culture media
Document and Ref: A study by Zhu et al demonstrate that extended culture before cryopreservation allows for selection of viable embryos with a high chance of implantation after warming. The high blastocyst formation rate (73.8%) using Vitrolife culture media, together with an efficient cryopreservation program resulting in survival rates of >96% should eliminate any earlier concerns about blastocyst culture and cryopreservation.

Scientific summaries
English
Embryo culture in G-Series results in significantly higher cell numbers and implantation rates compared to culture in HTF
Document and Ref: A study by Kleijkers show that besides cell numbers and implantations rates, gene expression and certain pathways in preimplantation embryos are also affected by culture medium.

Scientific summaries
English
G-Series culture media improve blastocyst development
Document and Ref: Data from two studies presented at Annual Meeting of the Japanese Society of Clinical Embryologist show that good quality embryo rate and blastocyst development can be improved when using G-Series culture media (G-IVF, G-1 and G-2) from Vitrolife compared to a HTF-based non-hyaluronan containing media.

Scientific summaries
English
G-Series culture media shown to increase blastocyst development compared to control
Document and Ref: In a study by Matsuoka et al, embryo culture was done either Vitrolife’s G-Series™ culture media or a mix of sequential culture media (control). The results show that good-quality blastocyst development was more frequent after culture in G-Series media containing hyaluronan.

Short protocols
English
G-Series™ ICSI procedure
Document and Ref: REF 19122. Version 02. Date of issue 220923. This short protocol describes how to perform ICSI using ICSI™, HYASE™-10x, OVOIL™ and G-Series medium G-MOPS™ PLUS .

Short protocols
English
G-Series™ Embryo culture G-1 PLUS and G-2 PLUS
Document and Ref: REF 19183. Version 07. Date of issue 220923. This short protocol describes how to culture embryos from after fertilisation until the blastocyst stage using the G-Series™ sequential media G-1™ PLUS, G-2™ PLUS and OVOIL™.

SSCP
English
SSCP G-1 PLUS
Document and Ref: REP-3354. Version 6.0. 2025/03/10
Do you have a question about this product?
Contact us
Find your local Vitrolife representative
Contact usMaximise success every step of the way
We provide what you need to secure improved results throughout the IVF journey.