ICSI™
ICSI made safe, using recombinant human albumin.
Description Viscous sperm handling solution containing recombinant human albumin (10 mg/ml) and PVP.
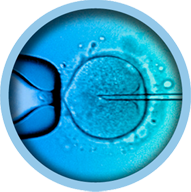
Purpose For the immobilisation and isolation of sperm prior to intracytoplasmic sperm injection, ICSI.
Application For use after pre-equilibration at +20 ± 5ºC and ambient atmosphere.
Storage Store dark at +2 to +8°C
Recombinant albumin for safety
ICSI is a viscous sperm handling solution designed to facilitate sperm immobilization and injection into the oocyte. It contains recombinant human albumin for safety and the most widely used molecule used for this purpose, polyvinylpyrrolidone or PVP. PVP is an inert molecule that does not take part in any metabolic pathway.

From scientific breakthrough to standard procedure
In 1992 science brought hope to thousands of childless couples when the first pregnancies generated by ICSI were reported. This meant that male infertility factor was less of an obstacle to starting a family.
At its introduction in the early 1990s, fertilisation rates after ICSI were around 30%.1 Thanks to further modifications of the procedure, fertilisation rates of over 80% are now possible and ICSI is now used as the method for fertilisation in around 65% of all treatment cycles globally.
Support Documents
Documents Language

Package inserts
English
Package insert ICSI - Multi-language
Document and Ref: REF 20430, version 10

Safety data sheets (SDS)
English
SDS ICSI
Document and Ref: Document and Ref: Date Revised : 04/16/2025.Revision No : 7. Safety data sheet for ICSI

SSCP
English
SSCP ICSI
Document and Ref: REP-4296. Version 6.0. 2025/04/10

Short protocols
English
G-Series™ ICSI procedure
Document and Ref: REF 19122. Version 02. Date of issue 220923. This short protocol describes how to perform ICSI using ICSI™, HYASE™-10x, OVOIL™ and G-Series medium G-MOPS™ PLUS .

Short protocols
English
Gx-Series™ ICSI procedure
Document and Ref: REF 19179. Version 1. Date of issue 220923 This short protocol describes how to perform ICSI using ICSI™, HYASE™-10x, OVOIL™ and Gx-Series medium Gx-MOPS™ PLUS and Gx-TL™
Do you have a question about this product?
Contact us
Find your local Vitrolife representative
Contact usMaximise success every step of the way
We provide what you need to secure improved results throughout the IVF journey.