eWitness – Reliable Electronic Witnessing System for IVF Labs
Automating IVF with Confidence to protect patients and staff from the risks of errors.
Disclaimer: eWitness is currently not available in all markets. Please contact our representatives in your area for more information.

Optimise Lab workflow and enhance security with reliable insights
eWitness brings automation, accuracy, and trust to every step of the IVF process, safeguarding compliance and peace of mind for embryologists, lab managers, and patients across the entire workflow
Discover the Latest eWitness Release: New Features and Enhanced Functionality for Streamlined, Error-Free IVF Workflow
The latest eWitness release introduces seamless EmbryoScope integration, a customizable KPI dashboard, photo capture for documentation, flexible witness and overrule options, enhanced GDPR compliance, release note pop-ups, and customisable misscan reasons to streamline and safeguard IVF lab workflows.
Learn more about new features and enhanced functionalityAdvancing IVF Treatment process quality
The IVF treatment process has advanced rapidly in recent years, with new technologies adding both opportunity and complexity. As procedures become more intricate, safeguarding every step has become more critical than ever.
eWitness is an error prevention system designed specifically for IVF. By scanning, registering, and validating each action, it ensures full traceability and oversight.
This helps prevent critical errors such as the mix-up of sperm, eggs, or embryos and gives clinics confidence that the entire workflow is secure, accurate, and free from human error.

Explore the advantages of choosing eWitness
eWitness is a flexible, user-friendly witness system designed to seamlessly fit into any IVF clinic. By automating daily workflow processes, eWitness enhances operational efficiency and minimizes manual workload, all while increasing reliability and quality through electronic double-checking of every action. Implementation is straightforward, requiring no workplace adjustments and only minimal hardware additions to your lab. Once configured for your clinic's workflow, eWitness guides users through every step of the fertility process with intuitive support. The label printer produces individual labels to prevent waste, and all printed labels and RFID tags are compliant with MEA standards, ensuring safety and quality throughout IVF procedures.

.

Easy implementation

Optimised Workflow: Flexibility, Efficiency, and Time Savings

Safety and Compliance

When compairing At UZ Gent, eWitness brings safety to our lab processes, as it results in an undisturbed, calm and focussed work environment for my team. eWitness is also connected to IDEAS, our EMR, resulting in a streamlined process that enhances overall process efficiency, all thanks to our effective partnership with the eWitness team.
- Dr. K. Tilleman, Director IVF Lab at UZ Gent Belgium
Built-in benefits
Seamless integration, dependable performance, and the flexibility to grow with your lab’s needs.
- High efficiency during day-to-day practice
- Reduced workload & peace of mind
- Quality assurance – including batch tracking (material registrations)
- Outstanding reliability through error reduction
- Closed loop registration through the use of one system
- Detailed insights through data
- Simple installation procedure

Effective safety, made simple with eWitness.

3 Easy Steps:
Scan, record and validate
eWitness logs each step in the fertility process by scanning the label with a barcode scanner and/or RFID reader. The patient identity and patient material are registered for validation. Regardless of the location or procedure, quality and progress are assured within seconds.
Explore What Makes eWitness Unique
eWitness is more than a electronic witnessing solution, it’s a comprehensive platform designed to enhance safety, efficiency, and compliance in every step of the IVF laboratory process. Discover the core components and advanced features that empower your team to deliver the highest standard of care with confidence.
eWitness Management System
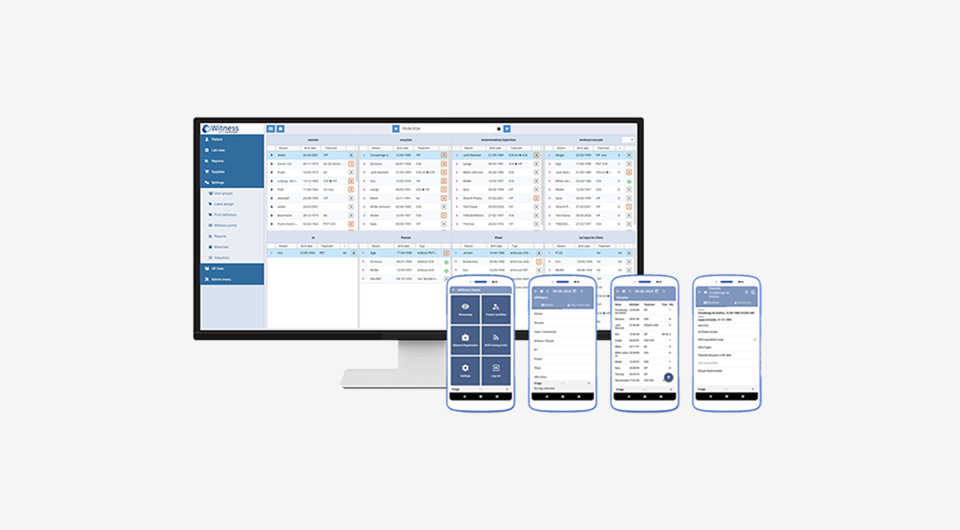
eWitness Management System is a web-based application that enables you to create your own procedures and witness points, design your own labels, create your KPI reports and manage the users accounts. Furthermore, eWitness provides a dashboard with a complete overview of all the patients, the total workload of the day and insight in the logging of the witness points.

Handheld scanner
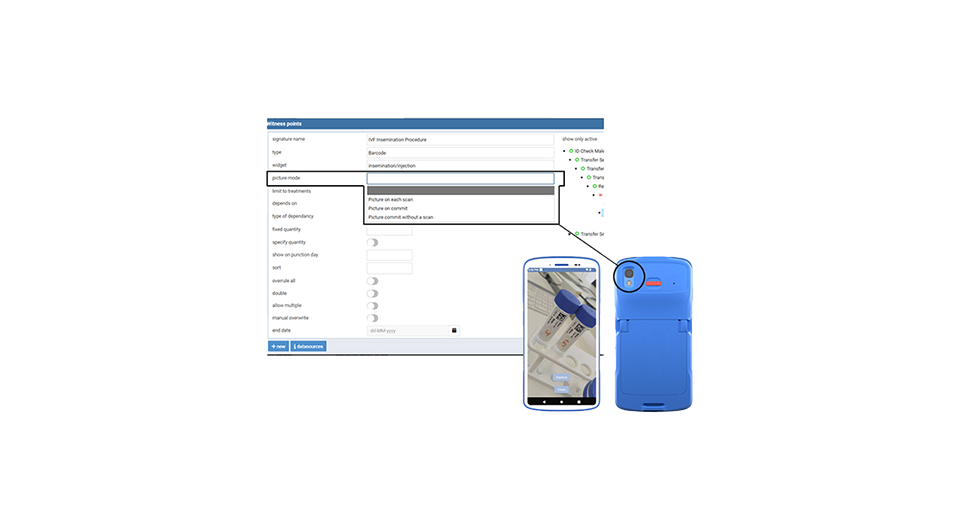
The system uses flexible and wireless handheld scanners. This allows you to securely scan barcodes on the label at any time anywhere and register the step in eWitness.

RFID reader
eWitness works with flexible and hands-free RFID readers. It is not necessary to replace flow cabinets as the eWitness RFID readers are easy to add to your existing environment. This ensures a fast and cost-effective implementation.

Labels and label printers
Each unique eWitness label contains all required patient identification data, eliminating handwritten and blind labels to reduce errors. Clinics can customise labels, which are available as cryo-resistant, standard barcode, or RFID formats (for non-cryo use). All labels are MEA tested and embryo safe for optimal safety and compliance. eWitness supports IVF workflows with traditional barcode, RFID, and cryo-resistant labels, all printable with the same versatile printer to simplify operations and reduce equipment needs.
New Features and Enhanced Functionality in eWitness
Streamline Your Workflow with eWitness and EmbryoScope
eWitness and EmbryoScope™ integration addresses common workflow challenges in the laboratory by automating patient registration and synchronizing critical data between systems. Patient information is seamlessly exchanged between eWitness and EmbryoScope, enabling quick selection, barcode generation within EmbryoViewer, and traceable dish labelling in eWitness. This streamlined workflow reduces manual input errors and ensures complete traceability for audit-ready laboratory management.
Register now for updates and early access!

KPI Dashboard
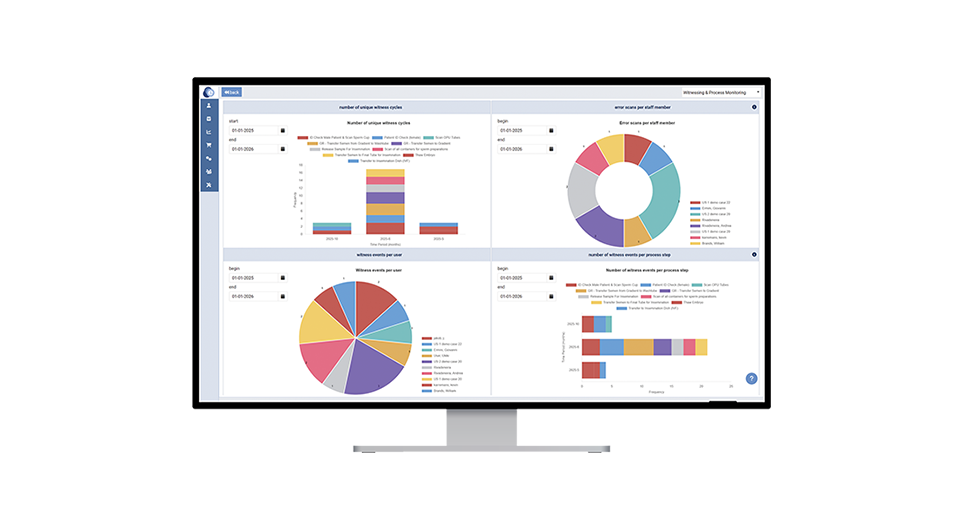
eWitness collects data on all actions, such as witnessing or batch tracking. The KPI Dashboard consolidates this information into a simple, customisable interface, enabling quick access to metrics for performance monitoring and benchmarking across teams and sites. The dashboard can be tailored to your needs, provides continuous insight for proactive management, supports the development of preventative protocols, and allows you to easily download detailed and customisable reports.
Photo Capture
Photo Capture enables automatic image documentation at every key process step, securely linking photos to each event record. This enhanced visual documentation delivers clear traceability, supports compliance, and provides clear visual proof of best practice. By making every action transparent and auditable, eWitness helps reduce risk, resolve questions with confidence, and uphold the highest standards of patient care, without disrupting workflow.

eWitness + square labware dishes
Optimal traceability, effortless barcode scanning, and secure sample management
- Seamlessly compatible label placement on every dish—no manual adjustment needed.
- Effortless barcode scanning and clear, human-readable content.
- Prevents blocked barcodes or lid issues for smooth lab operation.
- CE-marked for IVF: supports full traceability and regulatory compliance.
- Reduces errors and increases sample tracking confidence.
Learn more
With these new features and enhanced functionalities, eWitness makes error-free, streamlined IVF workflows and regulatory peace of mind more achievable than ever.
Contact us for more information or a personalised product demonstrationTrusted by IVF Professionals Worldwide
Hear directly from embryologists, lab managers, clinics, and sperm donor banks who rely on eWitness every day. Their feedback not only guides our solution’s ongoing development, but it also demonstrates how eWitness consistently delivers reliable safety, unmatched transparency, and genuine peace of mind.
.

“eWitness reduces the chance of errors and provides structure in our fertility lab. It contributes greatly to our quality assurances during the IVF treatment.”
Dr. E. Slappendel, clinical embryologist Nij Geertgen the Netherlands, part of FutureLife group

“We are very pleased with both the system and the services. Our laboratory staff and clinics benefit from an added layer of security provided by the electronic witnessing system. The system is user-friendly, and the biometric (fingerprint) identification of donors has been very well received by our donors.”
Maja Døvling Kaspersen Fedder, Born Donor Bank, Denmark

Webinar: 'Safety of your gametes and embryos with eWitness!'
In this webinar Dr. Kellt Tilleman share and demonstrate the implementation and use of an electronic witnessing system in their IVF laboratory to ensure the safe tracking.
eWitness is currently not available in all markets. For the US market, barcode labeling is available and other labeling and tracking systems will be introduced in 2025. This live webinar was held in October 2024.
Watch webinarFAQ
General Features
-
How easy is eWitness to use?
eWitness is designed to be user-friendly and intuitive. Comprehensive training is provided to help you navigate the system efficiently.
-
Does using eWitness add time to our workload, especially to each procedure?
eWitness significantly reduces staff time. It takes just a few seconds for one user to witness each procedure, saving time that would otherwise be spent on double-checking, completing witnessing forms, writing on dishes, interrupting colleagues, waiting for availability, transposing data from paper forms into software, auditing, analysing data, and producing reports. Gametes and embryos spend no more time outside the incubator than they currently do, and usually for significantly less time.
-
Is it available in multiple languages?
Yes. Currently available in English, Dutch, Danish, French, German, Greek, Italian, and Spanish.
Compatibility and Labware
-
Which of our labware (Plastics) are eWitness labels compatible with?
All labware. Our eWitness system includes various shapes and sizes to accommodate all labware from all known manufacturers, including vitrification devices, biopsy tubes and square labware dishes.
-
How do square labware dishes enhance eWitness?
Vitrolife square labware dishes. Square labware dishes not only prevent blocked barcodes or lid issues for smooth lab operation but also reduceerrors and increase confidence in sample tracking, supporting full regulatory compliance (CE-marked for IVF).
-
Can eWitness be used in a specific chamber in our incubator, or a specific cane within our cryostores?
Yes, these locations can be barcoded with the patient's name, or an external barcode can be assigned to a location or piece of apparatus if these items already have a permanent barcode attached.
Implementation and Integration
-
How long will it take us to get eWitness implemented?
The eWitness system can typically be implemented to Phase 1 level within 12 weeks from placing your order, provided we have engagement from your superusers, users, and IT department. Phase 2 implementation (integration with the end user's IT network and/or an EMR if ordered) will proceed in parallel and may take longer, depending on the engagement level from the IT department and EMR provider, and whether it is an EMR we have previously integrated with.
-
Can eWitness link with our existing EMR (Electronic Medical Records) database?
Yes, the eWitness database can be linked with an EMR via an API connection. This includes basic patient information to avoid data entry duplication, saving time and reducing the risk of transcription errors. Our Service & Support Team can assist your IT department/EMR provider in establishing this connection.
-
Is full bi-directional integration with ES a unique capability of eWitness?
Yes, eWitness supports true bidirectional integration with ES, facilitating data sync between eWitness and enterprise platforms.
Pricing, Technology and Support
-
How much will eWitness cost us?
There is a license fee for each patient treatment cycle for the software. Higher annual cycles result in lower fees per cycle. Next to that there is a yearly fee for the hardware. Alternatively, you can purchase the equipment to lower the yearly fees. Supplies like barcode labels and RFID tags can be ordered as needed. The price can be calculated for by eFertility by filling in a formular. Complete the following form to request a quote:
-
Is RFID more expensive than barcodes?
Yes, RFID tags are more expensive compared to barcode labels but l less expensive to many other competitors.
-
Can barcodes and RFID be used together?
Yes, you can start with barcode eWitness and add RFID later if desired. In situations where it is appropriate, you may also scan items with a handheld scanner, this is especially beneficial and is possible after the patient has been linked to an RFID tag (typically performed the day before use). A key differentiator of our solution is that the patient's name and ID are printed directly on the RFID label. Competing systems, such as those using IR, require an additional printed label, this process is time-consuming, uses valuable space on dishes or tubes, and increases the risk of handling errors..
Saftey, Security and Unique features
-
Are the eWitness labels safe?
Yes, eWitness labels undergo MEA (Mouse Embryo Assay) tests to ensure safety. Next to that the ink stays on very well.
-
Are special labels used for cryo?
Yes, special labels are used for cryogenic storage. RFID labels are generally not suitable for cryo conditions because they can be affected by metal and extremely low temperatures. Instead, barcodes are typically applied to cryo straws, as they remain effective in such environments. However, using barcode labels requires an additional application step and may increase overall costs.
-
Can you use RFID for cryo?
No, we use special cryo labels because RFID cannot withstand the very low temperatures in the cryo vessel. Additionally, RFID cannot be read through metal, so the straw must be removed from the vessel for a short time. Therefore, we use barcodes on the straws.
-
Is Technical Support available?
Yes, technical support is available and can be reached using the contact details provided in the Service Level Agreement
Do you have a question about this product?
Contact us
Find your local Vitrolife representative
Contact usMaximise success every step of the way
We provide what you need to secure improved results throughout the IVF journey.