G-TL™
The first culture medium specifically designed for time-lapse.
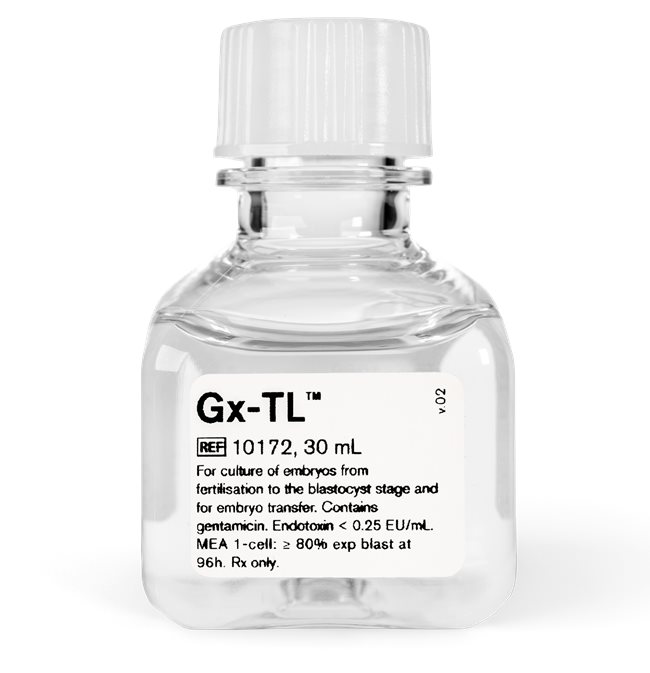
Description G-TL is a bicarbonate buffered medium containing hyaluronan and human serum albumin (5 mg/ml) for culture of embryos from fertilisation to the blastocyst stage and for transfer. Gx-TL has a unique combination of triple antioxidant protection for improved embryo viability.
Purpose Medium for culture of embryos from fertilisation to the blastocyst stage.
Application For use after pre-equilibration at +37°C and 6 % CO2.
Storage Store dark at +2 to +8°C
For truly undisturbed time-lapse culture
Time-lapse culture removes time limits and stresses from embryos during assessment while adding power to classic morphology. G-TL is a medium in the G-Series™ designed to optimise the use of time-lapse technology, allowing truly undisturbed time-lapse culture.
Gx-TL has a unique triple antioxidant protection to improve embryo viablity. Acetyl-L-Carnitine, Alpha-Lipoic Acidand N-Acetyl-L-Cysteine are working together to protect the embryo from oxidative stress.
Gx-TL with triple antioxidant protection
Reactive oxygen species are ubiquitous throughout any given IVF cycle. And they pose a significant threat to your embryo culture. To keep your embryos out of harm’s way, we have developed a unique triple antioxidant protection for improved embryo viability. The three antioxidants have been added to our culture media, Gx-TL, to form a protective barrier against oxidative stress.
Learn more about GX Media
Balancing stresses
G-TL provides uninterrupted culture conditions in an optimised culture environment to maximise embryo viability. The balanced mix of amino acids in G-TL is based on new knowledge about embryo metabolism and consumption of media constituents1. G-TL is designed to reduce ammonium load from breakdown products, while providing an optimised supply of amino acids for development.
G-TL has been shown to give high results when compared to sequential culture systems as well as monophasic culture systems.
Why you should use specific
embryo culture media for
specific applications
Watch this movie where Dr. Silversand describes the development of G-TL as well as present clinical results from a multicentre prospective randomised trial.
Watch movie

Time-lapse technology triggers new requirements on embryo culture media
The introduction of time-lapse technology has reduced the need of handling the embryo outside the incubator. Time-lapse technology has given a unique possibility to balance metabolic stress against less handling stress during embryo culture. Learn more in this blog post.
Read blog postStudy confirms high
performance of G-TL
Watch Dr. Hardarson present results from an international, prospective, controlled and randomised multicentre study evaluating G-TL in a clinical environment using time-lapse technology.
This lecture was a part of the Vitrolife Scientific Symposium at ESHRE 2014.

Support Documents
Documents Language

Package inserts
English
Package insert G-TL - Multi-language
Document and Ref: REF 26283, version 11

Package inserts
English
Package insert Gx-TL - Multi-language
Document and Ref: REF 26902, version 07

Safety data sheets (SDS)
English
SDS G-TL
Document and Ref: Document and Ref: Date Revised : 04/16/2025.Revision No : 4. Safety data sheet for G-TL

Safety data sheets (SDS)
English
SDS Gx-TL
Document and Ref: Document and Ref: Date Revised : 04/16/2025.Revision No : 3. Safety data sheet for Gx-TL

SSCP
English
SSCP G-TL
Document and Ref: REP-3361. Version 5.0. 2025/03/10

SSCP
English
SSCP Gx-TL
Document and Ref: REP-2364. Version 5.0. 2025/03/10

Scientific summaries
English
G-TL supports blastocyst development in large volume
Document and Ref: In a study presented at the ACE meeting in the UK, Shaw and co-workers compared single step time-lapse medium (Vitrolife, G-TL) with sequential series media (Sage, Quinn’s advantage) in large volume culture as well as micro drop culture. They found significantly higher day 5 blastocyst development rate in the G-TL group in both large and small volume culture.

Short protocols
English
G-Series™ G-TL Embryo Culture
Document and Ref: REF 19125 version 1. Date of issure 2019-08-12. This short protocol describes how to perform embryo culture using G-TL™, OVOIL™ and G-MOPS™ PLUS.

Short protocols
English
Gx-Series™ Gx-TL Embryo Culture
Document and Ref: REF 19181. Version 1. Date of issue 220923. This short protocol describes how to perform embryo culture using Gx-TL™, OVOIL™ and Gx-MOPS™ PLUS.

Short protocols
English
G-Series™ Blastocyst trophectoderm biopsy
Document and Ref: REF 19126. Version 02. Date of issue 220923. This short protocol describes how to perform blastocyst trophectoderm biopsy using OVOIL™ and G-Series™ media, G-2™ PLUS or G-TL™ and G-MOPS™ PLUS

Short protocols
English
Gx-Series™ Blastocyst trophectoderm biopsy
Document and Ref: REF 19180. Version 1. Date of issue 220923. his short protocol describes how to perform Blastocyst trophectoderm biopsy with OVOIL™, Gx-MOPS™ PLUS and Gx-TL™

Short protocols
English
G-Series™ ICSI procedure
Document and Ref: REF 19122. Version 02. Date of issue 220923. This short protocol describes how to perform ICSI using ICSI™, HYASE™-10x, OVOIL™ and G-Series medium G-MOPS™ PLUS .

Short protocols
English
Gx-Series™ ICSI procedure
Document and Ref: REF 19179. Version 1. Date of issue 220923 This short protocol describes how to perform ICSI using ICSI™, HYASE™-10x, OVOIL™ and Gx-Series medium Gx-MOPS™ PLUS and Gx-TL™
Do you have a question about this product?
Contact us
Find your local Vitrolife representative
Contact usMaximise success every step of the way
We provide what you need to secure improved results throughout the IVF journey.